8 Remarkable How To Find The Bond Order - The bond order equation is. The bond order for be2 is 0 (zero).
 How To Calculate Bond Order From Mo Diagram General . H, this means that only one pair of electrons
How To Calculate Bond Order From Mo Diagram General . H, this means that only one pair of electrons
How to find the bond order
13 Wonderful How To Find The Bond Order. Click to see full answer. How to find bond order using molecular orbital theory however, before determining the number of electrons in certain orbitals, one must fill these orbitals with electrons first. For example, for n≡n, the bond order is 3. How to find the bond order
Determining the bond order starting from a lewis structure is a task that can range from very easy to rather difficult. Bond order is an atomic level phenomenon, in which the elements share their electrons to fill their valance shell. What is the bond order of f2? How to find the bond order
Here are a number of highest rated o2 bond order pictures on internet. Example 1 nitrogen molecule the lewis structure shows six electrons shared between the two atoms. Here the bond order of co is consist of. How to find the bond order
We receive this nice of o2 bond order graphic. In simple terms, since f has 7 valence electrons thus by sharing of electrons with another f it forms a bond to fullfil its octate. Solved examples for bond order formula. How to find the bond order
It is defined among the number of bonding and antibonding electrons. No special bond order formula is usually required: In this video i will show you how to find bond order of any molecule just in seconds by a very simple trick.refer to this tableno. How to find the bond order
The bond order shows the number of chemical bonds present between a pair of atoms. You can also find its bond order using advance molecular orbital theory (mot). It has 3 resonating structures and total four bonds are present in one structure. How to find the bond order
A single bond has a bond order of 1, a double bond has a bond order of 2 and a triple bond has a bond order of 3. Suppose you are asked to find the bond order of hydrogen gas ( h 2). What is the bond order of f2?in simple terms, since f has 7 valence electrons thus by sharing of electrons with another f it forms a bond to fullfil its octate. How to find the bond order
The bond order is the quantity of bonding electron pairs between a pair of atoms. Q1] determine the bond order for hydrogen gas, h 2. It is a triple bond. How to find the bond order
Using advance molecular orbital theory (mot). You can also find its bond order using advance1. The higher the bond order, the more energy needed to break the bond. How to find the bond order
The valence shell of each beryllium atom is 2s2 so there are a total of four valence shell electrons for which we. I’ll tell you the shortest trick and really the shortest one to find bond order of almost any compound (please note that in some exceptional compounds this trick may not work but it works in 95% of the compounds We identified it from reliable source. How to find the bond order
Luckily, most cases you will encounter are the easy ones. In the article bond order formula, you have grasped the formulas to find the bond order based on molecular orbital theory and lewis structure. The lewis structure for hydrogen gas is as follows. How to find the bond order
You can explain the information conveyed by the bond order, such as stability, number No bond single bond double bond triple bond example 1: Considering this, what is the bond order of be2 −? How to find the bond order
And na is the number of electrons in the antibonding orbitals. Bond order refers to the general strength, or energy, of a bond. How to find the bond order can be illustrated by both theories. How to find the bond order
Bond order and bond length are two such important parameters that indicate the type and strength of bonds between a pair of atoms. The bond order formula can be defined as half of the difference between the number of electrons in bonding orbitals and antibonding To fill the orbitals, one must know the rules according to which orbitals are occupied. How to find the bond order
In a covalent bond between a pair of atoms, an individual bond has a bond order of 1 Also, it can bond two or more substance together in a compound. What is the average co bond order in the carbonate anion co32? How to find the bond order
What are bond order and bond length 2. The electronic configuration of hydrogen is (σ1s)2. For carbonate ion co3−2, the resonating structures are given as follows: How to find the bond order
Here are some examples of how to find bond order in molecules. Its submitted by giving out in the best field. A simpler way to find the bond order is to calculate the number of bonds between the atoms. How to find the bond order
The first step should always be to draw out your molecules. How to find the bond order
bond order DriverLayer Search Engine . The first step should always be to draw out your molecules.
 Calculating Bond Order YouTube . A simpler way to find the bond order is to calculate the number of bonds between the atoms.
Calculating Bond Order YouTube . A simpler way to find the bond order is to calculate the number of bonds between the atoms.
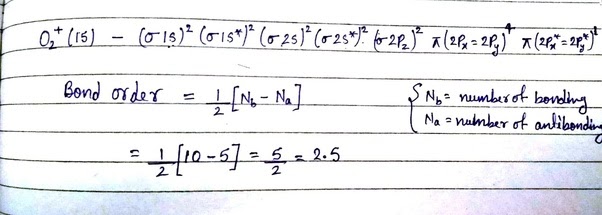
 How To Find Bond Order . Here are some examples of how to find bond order in molecules.
How To Find Bond Order . Here are some examples of how to find bond order in molecules.
 Bond Order of a compound Definition, Bond Order Formula . For carbonate ion co3−2, the resonating structures are given as follows:
Bond Order of a compound Definition, Bond Order Formula . For carbonate ion co3−2, the resonating structures are given as follows:
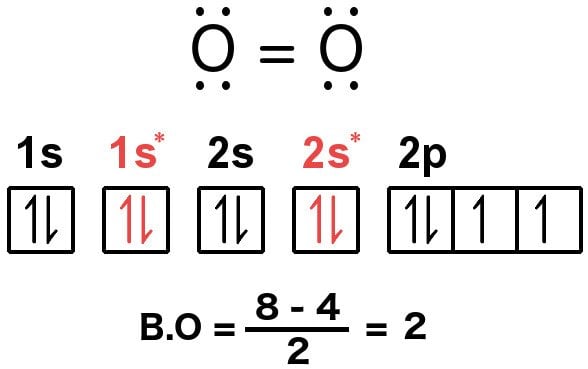
 bond order of 1.5 is shown by Brainly.in . The electronic configuration of hydrogen is (σ1s)2.
bond order of 1.5 is shown by Brainly.in . The electronic configuration of hydrogen is (σ1s)2.
Comments
Post a Comment